COVID-19 testing : Introduction, Types and Clinical Significance
Introduction of COVID-19 Testing
COVID-19 Testing is the most important tool for diagnosis of this coronavirus disease due to being similarities of various diseases. Coronavirus disease 2019 or COVID -19 is a viral disease and its causative agent is severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). It is a positive-sense single-stranded RNA virus. Antibodies ( IgM, IgG, and IgA) in blood and nucleocapsid protein (N protein) of the novel coronavirus and genes RdRP (RNA-dependent RNA polymerase ), E (envelope protein), N (nucleocapsid protein), ORF (open reading frame), and NSP ( nonstructural protein) and culture are diagnostic tools for laboratory diagnosis of SARS-CoV-2.
Types of COVID-19 Testing
- Serological method
- Molecular method
- Culture method
Serological method for COVID-19 Testing
Detection of antibodies
Part of the immune response to infection is the production of antibodies including IgM, IgG, and IgA. IgM can be a detectable infection in individuals starting 7 days and remaining IgG and IgA antibodies are usually detectable 14 days after the onset of the infection. The assay should perform in central laboratories and by point-of-care testing because of being virus under risk group 3 and highly contagious. The test can give a result in 15 minutes. It has a 91% clinical specificity rate and a 99% clinical sensitivity rate. The testing capacity is much faster than the conventional PCR assay of viral RNA.
It works under the principle of immunochromatography.
Test requirements
The test kit contains test device, droppers, buffer, and test manual
test sample ( blood/serum/plasma)
Timer
Procedure of IgM and IgG COVID-19 rapid test
- Collect blood/serum/plasma sample.
- Add a drop blood/serum/plasma (10µl) sample to sample well.
- Place 2-3 drops of buffer in the sample well.
- Read results after 15 minutes.
Result Interpretation
Negative Result: If only the quality control line (C) appears and G and M are not visible, then the result is negative.
Positive Result, M only: If both C and M appear, then the result is positive for the IgM antibody.
Positive Result, G only: If both C and G appear, then the result is positive for the IgG antibody.
Positive Result, G and M: If C and both G and M appear, then the result is positive for both the IgG and IgM antibodies
Test Invalid: No line at all or line/s either C or M or both but not on C. Repeat the test with a new test device.
- Molecular method for COVID-19 Testing
PCR tests
Using real-time reverse transcription-polymerase chain reaction (rRT-PCR), the test can run using respiratory samples like nasopharyngeal swab or nasal swab. Results are generally available within 4-6 hours. The RT-PCR test performed with throat swabs is only reliable in the first week of the disease whereas sputum is preferred for the second week because of the disappearing virus from the throat and it continues to multiply in the lungs. It looks for the “E” gene shared by all beta coronaviruses, and the RdRp gene-specific to SARS-CoV-2.
Testing algorithm varies manufacturer to manufacturer brochure but most common … go with this link @
On 21 March 2020, Cepheid similarly received EUA from the FDA for a test that takes about 45 minutes.
Other methods of viral detection
A test that uses isothermal nucleic acid amplification technology instead of PCR. Since this does not require a series of alternating temperature cycles this method can deliver positive results in as little as five minutes and negative results in 13 minutes.
A test that uses a monoclonal antibody that specifically binds to the nucleocapsid protein (N protein) of the novel coronavirus is being developed in Taiwan, with the hope that it can provide results in 15 to 20 minutes just like a rapid influenza test.
Culture method for COVID-19 Testing
The extremely rapid growth of the virus in culture
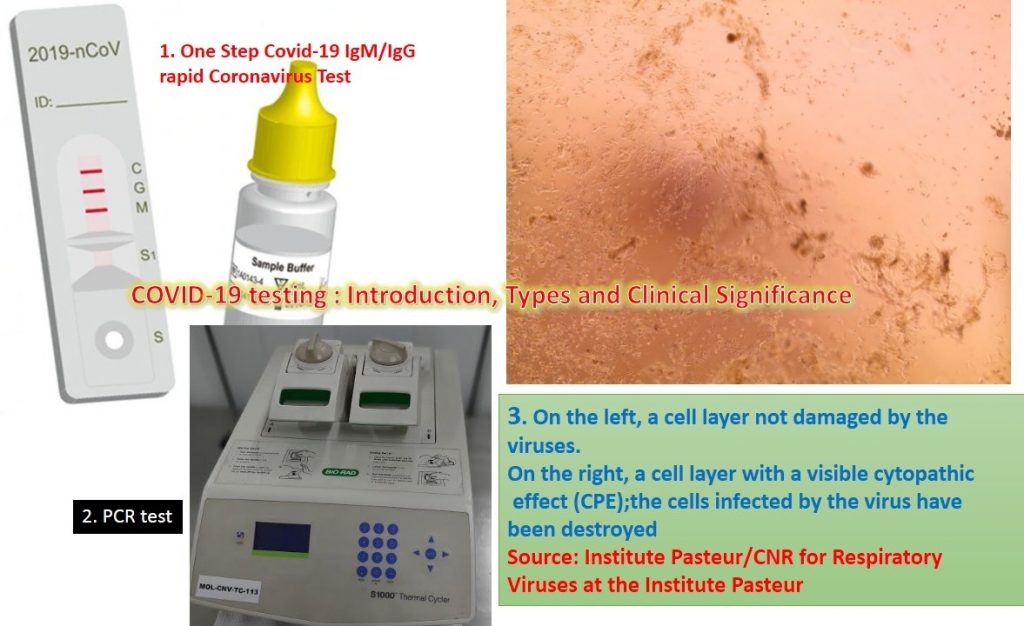
The Institute Pasteur was well prepared, and on Friday, January 24, 2020, the very day that the first cases were confirmed, it began the process of culturing the samples that had tested positive for the virus on Vero E6 cells. “Using detection methods, we had observed a high viral load in the samples taken from the patients in the hospital in Paris. This enabled us to identify which samples should be cultured first,” says Sylvie Behillil, Deputy Director of the CNR at the Institut Pasteur.
The viruses continued to be cultured over the weekend of January 25-26, 2020. By the morning of Monday, January 27, the culture had already grown! “We didn’t think that it would grow so quickly,” continues Sylvie Behillil. The rapid growth of the culture may be explained by “the high viral load in the samples,” but also by “the quality of the samples,” adds Vincent Enouf, Deputy Director of the CNR at the Institut Pasteur.
“We could see the cells becoming damaged and then grouping together, which can indicate that they have been infected. But we did not observe this cytopathic effect for all the inoculated samples; that reassured us that we had managed to isolate the strains, and this was then confirmed by additional analyses.”Virus 2019-nCoV now available for research
The National Reference Center (CNR) for Respiratory Viruses at the Institute Pasteur in Paris is one of WHO’s reference laboratories for coronavirus 2019-nCoV.
A total of eight people from the CNR and two from the P2M sequencing platform have been working on the virus this week and will continue to monitor the outbreak in France.
The isolation of coronavirus 2019-nCoV clears a vital hurdle for research, which has now begun. The Institut Pasteur immediately set up a task force to mobilize its experts with the aim of developing diagnostic, prevention, and treatment tools as quickly as possible to tackle the novel coronavirus.
Several Institute Pasteur teams are represented on the task force, which will focus its research on various scientific areas:
Understanding more about the virus and its pathogenesis;
Developing new diagnostic tools and searching for antibodies that may have therapeutic applications;
Vaccine development;
Epidemiology and modeling to develop outbreak control strategies.
Clinical significance of COVID-19 Testing
- Real-time reverse transcription-polymerase chain reaction (rRT-PCR) test is applicable for both screening and confirmatory of coronavirus RNA. This test is specific and is designed to only detect the RNA of the SARS-CoV-2 virus. It is used to confirm very recent or active infections.
- IgM can be a detectable infection in individuals starting 7 days and remaining IgG and IgA antibodies are usually detectable 14 days after the onset of the infection.
- Cultivation of SARS-CoV-2 needs trained staff with BSL3.
Further Readings
- Fellmann F. (March 2020). (in German) “Jetzt beginnt die Suche nach den Genesenen”. Tages Anzeiger. Retrieved 28 March 2020.
- “Letter from FDA”. FDA. 27 March 2020. Retrieved 2 April 2020.
- “2019 Novel Coronavirus (2019-nCoV) Situation Summary”. Centers for Disease Control and Prevention. 30 January 2020. Archived from the original on 26 January 2020. Retrieved 30 January 2020.
- “Curetis Group Company Ares Genetics and BGI Group Collaborate to Offer Next-Generation Sequencing and PCR-based Coronavirus (2019-nCoV)
- Testing in Europe”. GlobeNewswire News Room. 30 January 2020. Archived from the original on 31 January 2020. Retrieved 1 February 2020.
- Drosten, Christian (26 March 2020). “Coronavirus-Update Folge 22” (PDF). NDR. Archived (PDF) from the original on 31 March 2020. Retrieved 2 April 2020.
- “ABOUT US | NEWS”. www.kogene.co.kr.
- “Sunnyvale company wins FDA approval for first rapid coronavirus test with 45-minute detection time”. EastBayTimes.com. 21 March 2020. Archived from the original on 22 March 2020.
- www.sinica.edu.tw. Sinca. Retrieved 12 March 2020.
- https://eurekalert.org/pub_releases/2020-01/ip-ipi013120.php
- file:///C:/Users/Berry/Downloads/WHO-COVID-19-laboratory-2020.4-eng%20(1).pdf
