Hektoen Enteric Agar: Introduction, Principle, Composition, Preparation, Procedure, Colony Morphology, Uses and Keynotes
Introduction of Hektoen Enteric Agar
Hektoen enteric agar was developed in 1968 by Sylvia King and William I. Metzger in an effort to increase the recovery of Salmonella and Shigella species over the previously formulated Salmonella-Shigella (SS) Agar. This is also known as HEK and HEA. This medium is particularly useful in the isolation of Shigella species. They formulated HE Agar medium while working at the Hektoen Institute in Chicago and became able to increase the recovery of Salmonella and Shigella from clinical specimens. It is also a selective and differential medium used for the isolation and differentiation of gram-negative enteric pathogens.
(Himedia)
Ingredients Gms / Litre
- Protease peptone: 12.0
- Yeast extract: 3.0
- Lactose: 12.0
- Sucrose: 2.0
- Salicin: 9.0
- Bile Salts mixture (Equivalent to Bile Salt No. 3): 9.0
- Sodium chloride: 5.0
- Sodium thiosulfate: 5.0
- Ferric ammonium citrate: 1.5
- Acid fuchsin: 0.1
- Bromothymol blue: 0.065
- Agar: 14.0
- Distilled water: 1000 ml
Final pH 7.5±0.2
Principle of Hektoen Enteric Agar
The present formulation of HE Agar incorporates larger amounts of peptone in order to offset the inhibitory effect of bile salts. Also, sodium deoxycholate has been eliminated and the number of bile salts reduced. Bile salts allow for the selective nature of HE Agar by inhibiting gram-positive organisms. Bile salts can also be toxic for some gram-negative strains. Salicin, sucrose, and lactose are the fermentable carbohydrates present. They provide optimal differentiation of enteric pathogens. Lactose and sucrose, in increased concentration, aid in the differentiation of enteric pathogens from slow lactose fermenters. Bromothymol blue and acid fuchsin (Andrade’s) are added as acid-base indicators.
The addition of ferric ammonium citrate and sodium thiosulfate enables the detection of H2S, noted by the production of black-centered colonies. Sodium thiosulfate serves as the sulfur source while ferric ammonium citrate serves as the indicator. The remaining ingredients like yeast extract and proteose peptone provide nitrogen, vitamins, and amino acids. Agar acts solidifying agent and sodium chloride is the source of electrolytes whereas water is the source of hydrogen and oxygen.
Preparation of Hektoen Enteric Agar
- Suspend 72.66 grams of the powder of Hektoen Enteric Agar in 1 liter purified/distilled or deionized water.
- Mix thoroughly and heat with frequent agitation and boil for 1 minute to completely dissolve the powder.
- Avoid overheating and do not autoclave.
- Leave for cooling to 45-50°C.
- Mix well before dispensing.
- Pour into each plate and leave plates on the sterile surface until the agar has solidified.
- Store the plates in a refrigerator at 2-8°C.
Storage and Shelf life of Hektoen Enteric Agar
- Store in sleeve wrapping until just prior to use at 2-8ºC and away from direct light.
- Plates from opened stacks of 10 plates can be used for one week when stored in a clean area at 2 to 8° C.
- Media should not be used if there are any signs of deterioration (shrinking, cracking, or discoloration), contamination.
- The product is light and temperature-sensitive; protects from light, excessive heat, moisture, and freezing.
Test Requirements for Hektoen Enteric Agar
- Test specimens: Infectious material should be submitted directly to the laboratory without delay and protected from excessive heat and cold. If there is to be a delay in processing, the specimen should be inoculated onto an appropriate transport medium and refrigerated until inoculation.
- Hektoen Enteric (HE) Agar plates
- Inoculating loop
- Bunsen burner
- Incubator
- Control strains ( Enterococcus faecalis ATCC 29212, Escherichia coli ATCC 25922,Salmonella Typhi ATCC 6539, Shigella flexneri 12022 ATCC )
Test Procedure of Hektoen Enteric Agar
- Allow the plates to warm at room temperature, and the agar surface to dry before inoculating.
- Take Hektoen Enteric Agar plates.
- If material is being cultured directly from a swab, roll the swab over a small area of the surface edge.
- Streak the inoculum to obtain isolated colonies.
- A non-selective medium should also be inoculated. This increases the chance of recovery when the population of gram-negative organisms is low. It also provides an indication of other organisms present in the specimen.
- Incubate plates for 18-24 hours at 35ºC and protect from light.
- If negative after 24 hours, re-incubate for an additional 24 hours.
Result Interpretation of Hektoen Enteric Agar
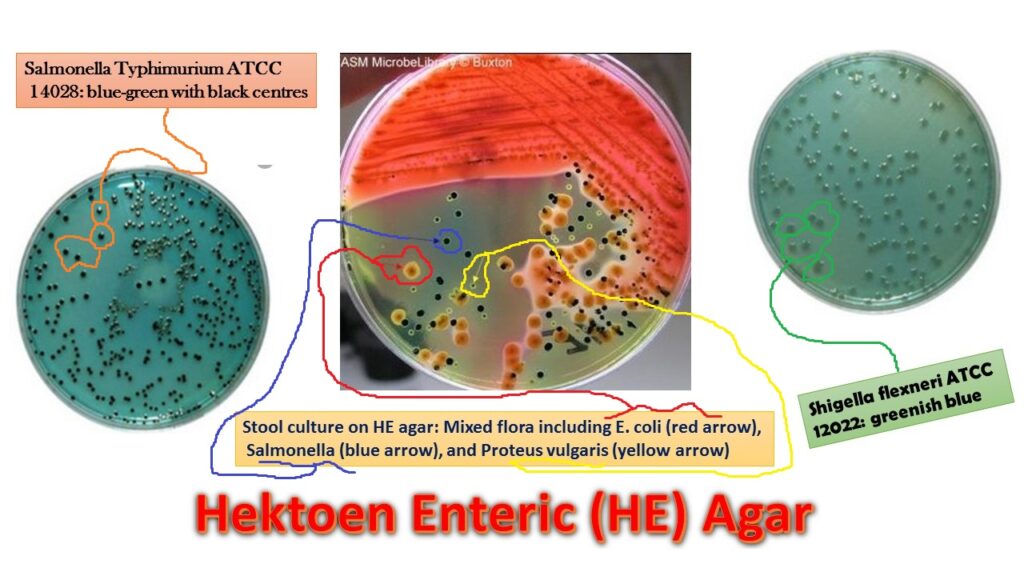
HE Agar is examined for typical colonial morphology after incubation. Fermentation of lactose, sucrose, or salicin results in the production of acid which gives rise to yellow-orange to salmon-colored colonies. Colonies of Salmonella and Shigella species are green to bluish-green in color. Salmonella species that produce H2S appear as blue-green colonies with black centers. H2S producers form black-centered colonies in the presence of ferric ammonium citrate and sodium thiosulfate
Colony characteristics of Hektoen Enteric Agar
- Salmonella Typhimurium ATCC 14028: blue-green with or without black centers
- Salmonella Abony NCTC6017: blue-green with or without
black centers - Salmonella Enteritidis ATCC 13076: blue-green with or without black centers
- Salmonella Typhi ATCC 6539: blue-green with or without black centers
- Escherichia coli ATCC 25922: orange (may have bile precipitate)
- Escherichia coli ATCC 8739: orange (may have bile precipitate)
- Shigella flexneri ATCC 12022: greenish blue
- Proteus species: strains are somewhat inhibited; colonies that develop are small transparent, and more glistening or watery in appearance than species of Salmonella or Shigella.
- Enterococcus faecalis ATCC 29212: Inhibited
- Staphylococcus aureus ATCC 6538: Inhibited
- Micrococcus luteus: Inhibited ( no growth)
Uses of HEA
- This medium is particularly useful in the isolation of Shigella species.
- It is also a selective and differential medium used for the isolation and differentiation of gram-negative enteric pathogens.
- HEA is currently using as a direct and indirect plating medium to recover gastrointestinal (GIT) pathogens, such as Salmonella and Shigella, from clinical (fecal samples) as well as non-clinical specimens (food and water) suspected of containing these organisms.
Limitations of Hektoen Enteric (HE) Agar
- Colony morphology is only presumptive identification and hence biochemical, immunological, molecular, or mass spectrometry testing be performed on colonies from pure culture for complete identification.
- The presence of antimicrobial agents in the specimen may delay or inhibit cultural growth and even additionally, antimicrobics may alter the characteristic appearance of the organism on the medium.
- It is recommended that selective enrichment broths like GN (Gram Negative) Broth or Selenite Cystine be used in conjunction with selective plating media for optimal isolation of enteric pathogens.
- Bile salts in the medium may crystallize over time and they appear as small spider-like puff-balls within the medium and but they do not affect performance.
- The color of HE Agar may shift from green to brownish during shipment. This is normal and should not affect the performance of the medium. Placing plates in refrigerated conditions (2-8°C) upon receipt overnight will result in the medium returning to a normal green appearance.
- Colonies of Proteus, which may or may not be inhibited, may resemble Salmonella or Shigella.
- The recovery of most Shigella and many Salmonella species from unpreserved stool specimens may be jeopardized if processing delays exceed 2-3 hours.
- HE Agar is currently recommended as one of several plating media for the culture of Enterobacteriaceae from stool specimens. This is due to its moderately selective nature as well as for its differentiation property.
Keynotes on Hektoen Enteric Agar
- Extra amounts of carbohydrates and peptones in this HEA are to offset the inhibitory effects of the bile salts.
- The two dyes, bromthymol blue, and acid fuchsin, of this medium, have lower toxicity than other dyes, and thus they enhance the pathogen recovery.
- HE agar is currently using as both a direct and indirect plating medium for fecal specimens to enhance the recovery of species of Salmonella and Shigella from heavy numbers of mixed normal fecal flora.
- A differential, selective medium for the isolation of Shigella and Salmonella species from enteric pathological specimens.
Precautions for Hektoen Enteric (HE) Agar
- This product may contain components of animal origin. Certified knowledge of the origin and/or sanitary state of the animals does not guarantee the absence of transmissible pathogenic agents and thus, it is recommended that these products be treated as potentially infectious, and handle observing the usual universal blood precautions. Do not ingest, inhale, or allow to come into contact with skin.
- This product is for in vitro diagnostic use only and it is to be used only by adequately trained and qualified laboratory personnel. Observe approved biohazard precautions and aseptic techniques. All laboratory specimens should be considered infectious and handled according to “standard precautions.
Further Readings
- Clinical Microbiology Procedure Handbook Vol. I & II, Chief in editor H.D. Isenberg, Albert Einstein College of Medicine, New York, Publisher ASM (American Society for Microbiology), Washington DC.
- Colour Atlas and Textbook of Diagnostic Microbiology. Editors: Koneman E.W., Allen D.D., Dowell V.R. Jr and Sommers H.M.
- Jawetz, Melnick and Adelberg’s Medical Microbiology. Editors: Geo. F. Brook, Janet S. Butel & Stephen A. Morse, 21st ed 1998, Publisher Appleton & Lance, Co Stamford Connecticut.
- Mackie and Mc Cartney Practical Medical Microbiology. Editors: J.G. Colle, A.G. Fraser, B.P. Marmion, A. Simmous, 4th ed, Publisher Churchill Living Stone, New York, Melborne, Sans Franscisco 1996.
- Textbook of Diagnostic Microbiology. Editors: Connie R. Mahon, Donald G. Lehman & George Manuselis, 3rd edition2007, Publisher Elsevier.
- https://en.wikipedia.org/wiki/Hektoen_enteric_agar
- https://catalog.hardydiagnostics.com/cp_prod/Content/hugo/HektoenEntericAgar.htm
- http://www.himedialabs.com/TD/MU467.pdf
- https://www.sciencedirect.com/topics/immunology-and microbiology /hektoen-enteric-agar
- http://www.oxoid.com/UK/blue/prod_detail/prod_detail.asp
- https://www.bd.com/resource.aspx?IDX=8970
