Hucker’s Modification Gram stain: Composition, Preparation and Procedure
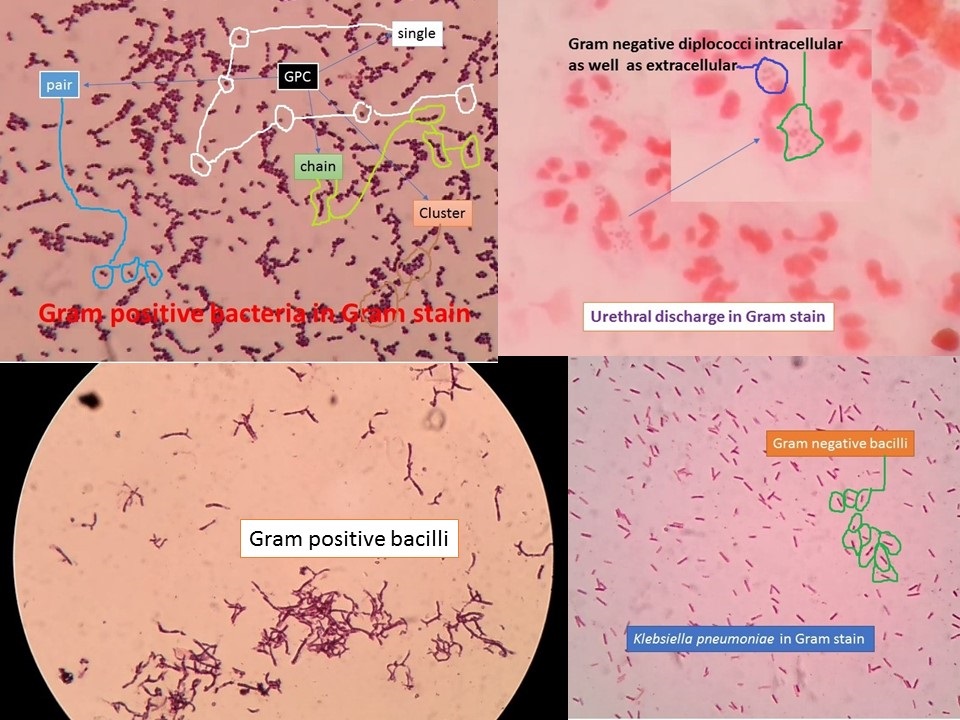
Introduction of Hucker Modification Gram stain
The Hucker modification Gram stain test was originally developed by Christian Gram in 1884. The modification currently used for general bacteriology was developed by Hucker in 1921. Gram-stain can be used effectively to divide all bacterial species into two large groups: those that take up the basic dye, crystal violet (Gram-positive), and those that allow the crystal dye to wash out easily with the Decolorizer alcohol or acetone (Gram-negative)
Hucker Modification Gram Stain and Reagent Preparation
Crystal violet solution (Hucker’s crystal violet)
Crystal violet stock solution
Crystal violet (90% to 95% dye content) 40 g
Ethanol, 95% 400 ml
Preparation
- Dissolve and mix in a glass bottle.
- Label with a 1-year expiration date, and stored at room temperature.
Ammonium oxalate solution (1%)
Ammonium oxalate (reagent grade) 16 g
Distilled water 1600 ml
Preparation
- Dissolve and mix in a brown glass bottle,.
- Label with a 1-year expiration date, and stored at room temperature.
Crystal violet working solution
Crystal violet stock solution 40 ml
Ammonium oxalate solution (1%) 160 ml
Preparation
- Filter crystal violet stock solution into a glass.
- Allow to filter completely, and then filter ammonium oxalate solution.
- Label with the earliest expiration date of stock solutions.
Gram’s iodine
Stock Lugol’s iodine solution
Iodine crystals (reagent grade) 25 g
Potassium iodide (reagent grade) 50 g
Distilled water 500 ml
Preparation
- Mix and let stand until dissolved in a brown glass bottle
- Label with a 6- month expiration date, and store at room temperature.
Sodium bicarbonate, 5% (w/v)
Sodium bicarbonate (reagent grade) 50 g
Distilled water 1000 ml
Preparation
- Dissolve in a glass bottle
- Label with a 1-year expiration date, and stored at room temperature.
Gram’s iodine
Stock Lugol’s iodine solution 60 ml
Distilled water 220 ml
Sodium bicarbonate (5%) 60 ml
Preparation
- Mix in a brown glass bottle.
- Label with a 6-month expiration date, and stored at room temperature.
Acetone-Alcohol Decolorizer
Acetone 500 ml
Ethanol (absolute) 475ml
Distilled Water 25 ml
Preparation
- Add to 25 ml D/W, 475 ml of absolute alcohol.
- Mix and transfer into a clean bottle.
- Then add immediately, 500 ml acetone to the bottle and mix well.
Safranine (Counter Stain)
Safranine 10.0 gm
Distilled Water 1000 ml
Preparation
- Weigh 10 gm of Safranine in a clean piece of paper, and transfer to a clean bottle.
- Then add 1-liter D/W to the bottle and mix well until Safranine dissolves completely.
Gram Staining Procedure of Hucker Modification Gram stain
The following steps are involved in Gram stain:
- Prepare and dry a thin film of the material to be examined.
- Heat fix the material on the slide and allow it to cool before staining.
- Flood the slide with a crystal violet stain and allow it to remain without drying for 30 to 60 seconds.
- Rinse the slide with tap water, shaking off excess.
- Flood the slide with iodine solution and allow it to remain on the surface without drying for twice as long as the crystal violet is in contact with the slide surface.
- Rinse the slide with tap water, shaking off excess.
- Flood the slide with acetone alcohol decolorizer for 10 seconds and rinse immediately with tap water until no further colors flow from the slide with the decolorizer. Thicker smear requires more aggressive decolorizing.
- Flood the slide with counterstain (Safranine) for 30 seconds and wash off with tap water.
- Blot the slide between two clean sheets of blotting paper and examine microscopically under oil immersion at 100X.
Further Readings
- Bailey & Scott’s Diagnostic Microbiology. Editors: Bettey A. Forbes, Daniel F. Sahm & Alice S. Weissfeld, 12th ed 2007, Publisher Elsevier.
- Clinical Microbiology Procedure Handbook Vol. I & II, Chief in editor H.D. Isenberg, Albert Einstein College of Medicine, New York, Publisher ASM (American Society for Microbiology), Washington DC.
- Colour Atlas and Textbook of Diagnostic Microbiology. Editors: Koneman E.W., Allen D.D., Dowell V.R. Jr, and Sommers H.M.
- Jawetz, Melnick and Adelberg’s Medical Microbiology. Editors: Geo. F. Brook, Janet S. Butel & Stephen A. Morse, 21st ed 1998, Publisher Appleton & Lance, Co Stamford Connecticut.
- Mackie and Mc Cartney Practical Medical Microbiology. Editors: J.G. Colle, A.G. Fraser, B.P. Marmion, A. Simmous, 4th ed, Publisher Churchill Living Stone, New York, Melborne, Sans Franscisco 1996.
- Textbook of Diagnostic Microbiology. Editors: Connie R. Mahon, Donald G. Lehman & George Manuselis, 3rd edition2007, Publisher Elsevier.
