Bacterial Coagulase Test: Introduction, Principle, Procedure, Result Interpretation, Limitations and Keynotes
Introduction of Bacterial Coagulase Test
The bacterial coagulase test is an enzymatic test that is used to differentiate Staphylococcus aureus (positive) from coagulase-negative staphylococci (CoNS). Coagulase is an enzyme produced by S. aureus that converts soluble fibrinogen of plasma to insoluble fibrin. S. aureus produces two forms of coagulase i.e. bound and free coagulase.
- Slide coagulase test is proceeded to detect bound coagulase or clumping factor.
- A tube coagulase test is performed to detect free coagulase.
Principle of Bacterial Coagulase Test
Staphylococcus aureus is the most pathogenic species among staphylococci. It is different from other species by the presence of coagulase. This enzyme is a thermostable thrombin-like substance that activates fibrinogen to form fibrin, resulting in a fibrin clot. This is demonstrated in the test tube by the formation of a clot when plasma is inoculated with Staphylococcus. The substance is known as free coagulase ( detectable in tube coagulase test) since it is liberated by the cell. In most, but not all, S. aureus organisms, fibrinogen binding cell surface receptor is also present in the cell wall, called bound coagulase’ or clumping factor ( detectable on slide coagulase test). The clumping factor is demonstrated by the ability of the organism to act directly on the fibrinogen in the plasma to clump it in a slide assay. The test for clumping factor is rapid but requires several colonies and this factor is not present in all S. aureus organisms. In addition, the clumping factors can be masked by cell surface capsular polysaccharides and can be present in other species, namely, Staphylococcus lugdunensis and Staphylococcus schleiferi and the reason, negative slide tests must be followed with a confirmatory tube test.
Requirements for Bacterial Coagulase Test
Test organism
Colonies of gram-positive cocci in clusters that are catalase-positive, as part of the identification of S. aureus.
Plasma
Note: Rabbit plasma is preferred rather than human plasma due to the following reasons:
- Human plasma for the test is less sensitive.
- It is potentially infectious with human pathogenic viruses.
Other accessories
Loops or sterile sticks
Glass or plastic tubes
clean and grease-free glass slides or black coated cards
Quality control strains
Positive Control (PC): Staphylococcus aureus ATCC 25923
Negative Control (NC): Staphylococcus epidermidis ATCC 12228
Procedure for slide Coagulase Test Method
It is for the detection of bound coagulase.
- Put a drop of distilled water on each end of a slide or you can also put it on two separate slides.
- Emulsify a colony of the test organism (To avoid misidentifications, only perform this test on classic-looking white to yellow, creamy, opaque, hemolytic colonies of gram-positive cocci in clusters that are catalase-positive. Hemolysis should only be observed on a fresh blood agar plate at equal and less than 18 hours) in each of the drops to make two thick suspensions. Note: Avoid taking colony from mannitol salt agar culture because of not suitable for coagulase testing. The organism must first be cultured on nutrient agar or blood agar.
- Add just a loopful of plasma to one of the suspensions, and mix gently.
- Look for clumping of the organisms within 10 seconds, No plasma is added to the second suspension.
- This is used to differentiate any granular appearance of the organism from true coagulase clumping or agglutination.
Procedure for Tube Coagulase Test Method
- Take 0.5 ml of plasma which is diluted 1 in 5 with saline. If there is stored plasma, bring it to room temperature (25°C).
- Inoculate with one colony of staphylococcus growing on a non-inhibitory medium.
- Incubate the tube at 37°C without CO2 for up to 4 hours and observe for clot formation hourly. Do not agitate the tube during observations; rather, gently tilt to observe the clot.
- If the clot is not observed at that time, reincubate the tube at room temperature and read after 20 hours.
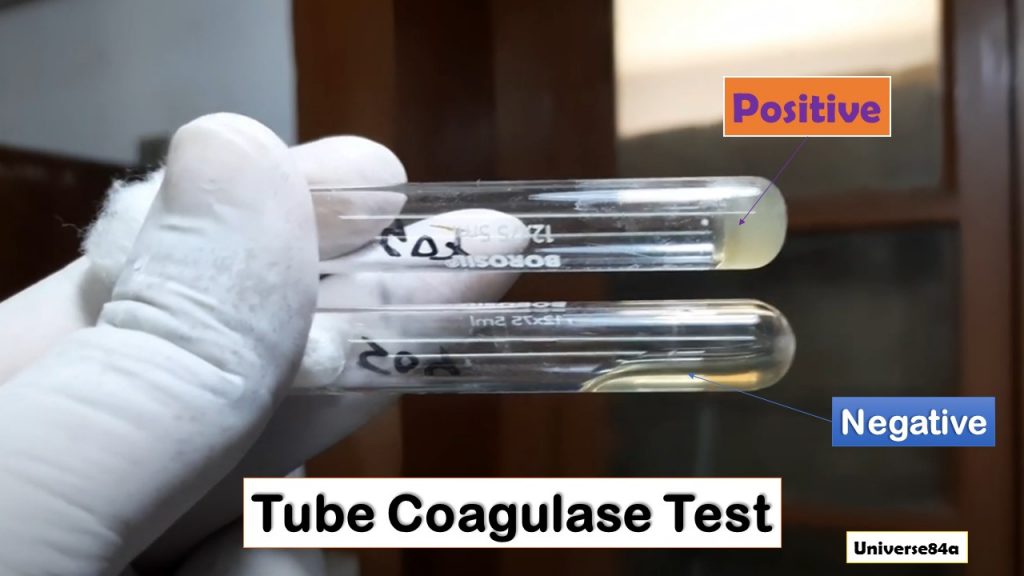
Observations of Bacterial Coagulase Test
In a positive slide test, evoke clumping of the organism shows the presence of the bound coagulase whereas in a positive tube test, the plasma in the tube clot and does not flow when the tube is inverted. One thing we should know; on continued incubation, the clot may be lysed by fibrinogen secreted by some strains.
Result and Interpretation of Bacterial Coagulase Test
For slide coagulase test
Positive: Clumping or agglutination within 10 secs
Negative: No clumping within 10 secs
For tube coagulase test
Positive: Clot formation
Negative: No clot formation
All strains producing negative slide tests must be tested with the tube coagulase test. If there is any degree of the clot, the test should consider positive.
List of coagulase-positive staphylococci
- Staphylococcus aureus
- Staphylococcus schleriferi
- Staphylococcus felis
- Staphylococcus intermedius
- Staphylococcus lutrae
- Staphylococcus hyicus
- Staphylococcus lodgunensis
Note: among them only slide positive organisms-
- Staphylococcus lodgunensis
- Staphylococcus schleriferi
whereas only tube coagulase test positive-
- Staphylococcus hyicus
Limitations of Coagulase Test
- Coagulase testing cannot be performed from growth on mannitol salt agar.
- Methicillin-resistant S. aureus (MRSA) can be deficient in bound coagulase, which results in a negative slide test.
- S. intermedius and S. hyicus may be positive in the tube test.
- Do not use citrated blood, as false-positive results can occur.
Keynotes on Bacterial Coagulase Test
- Do not practice leaving the test at 37°C for more than 4 hours, since S. aureus fibrinolysin can lyse the clot.
- If 4 hours of incubation at 37°C is inconvenient for staffing, the test is most sensitive when incubated at room temperature(25°C) for the entire time, but the clot may take longer to form.
- Alternatively, if the tube is left at 37ºC, add a drop or two of 5% CaCl2 at 24 h. If a clot forms, the isolate is coagulase-negative; if a clot does not form, fibrinolysin has lysed a previously formed clot and this result confirms the isolate as coagulase positive but the reference for this article is unpublished data of H. D. Isenberg, editor in chief Clinical Microbiology Procedures Handbook, original and second editions.
- The coagulase clot can be destroyed by S. aureus fibrinolysin or staphylokinase, a plasmid-carried enzyme which is more active at 35°C than at 25°C.
Further Readings
- Cowan & Steel’s Manual for identification of Medical Bacteria. Editors: G.I. Barron & R.K. Felthani, 3rd ed 1993, Publisher Cambridge University Press.
- Bailey & Scott’s Diagnostic Microbiology. Editors: Bettey A. Forbes, Daniel F. Sahm & Alice S. Weissfeld, 12th ed 2007, Publisher Elsevier.
- Clinical Microbiology Procedure Handbook Vol. I & II, Chief in editor H.D. Isenberg, Albert Einstein College of Medicine, New York, Publisher ASM (American Society for Microbiology), Washington DC.
- Colour Atlas and Textbook of Diagnostic Microbiology. Editors: Koneman E.W., Allen D.D., Dowell V.R. Jr, and Sommers H.M.
- Jawetz, Melnick and Adelberg’s Medical Microbiology. Editors: Geo. F. Brook, Janet S. Butel & Stephen A. Morse, 21st ed 1998, Publisher Appleton & Lance, Co Stamford Connecticut.
- Mackie and Mc Cartney Practical Medical Microbiology. Editors: J.G. Colle, A.G. Fraser, B.P. Marmion, A. Simmous, 4th ed, Publisher Churchill Living Stone, New York, Melborne, Sans Franscisco 1996.
- Textbook of Diagnostic Microbiology. Editors: Connie R. Mahon, Donald G. Lehman & George Manuselis, 3rd edition2007, Publisher Elsevier.
